For students, think of it as how eager an element is to form a compound.Įlectronegativity is especially useful in combination with the octet rule. Why? Because there is a direct relationship between how full sub-shells are and how electronegative an element is. Electronegativity, which is the sharing of electrons in a bond between atoms, is predictable based on the location of an element in the Periodic Table.NB: (Sub-)shells will be discussed below. 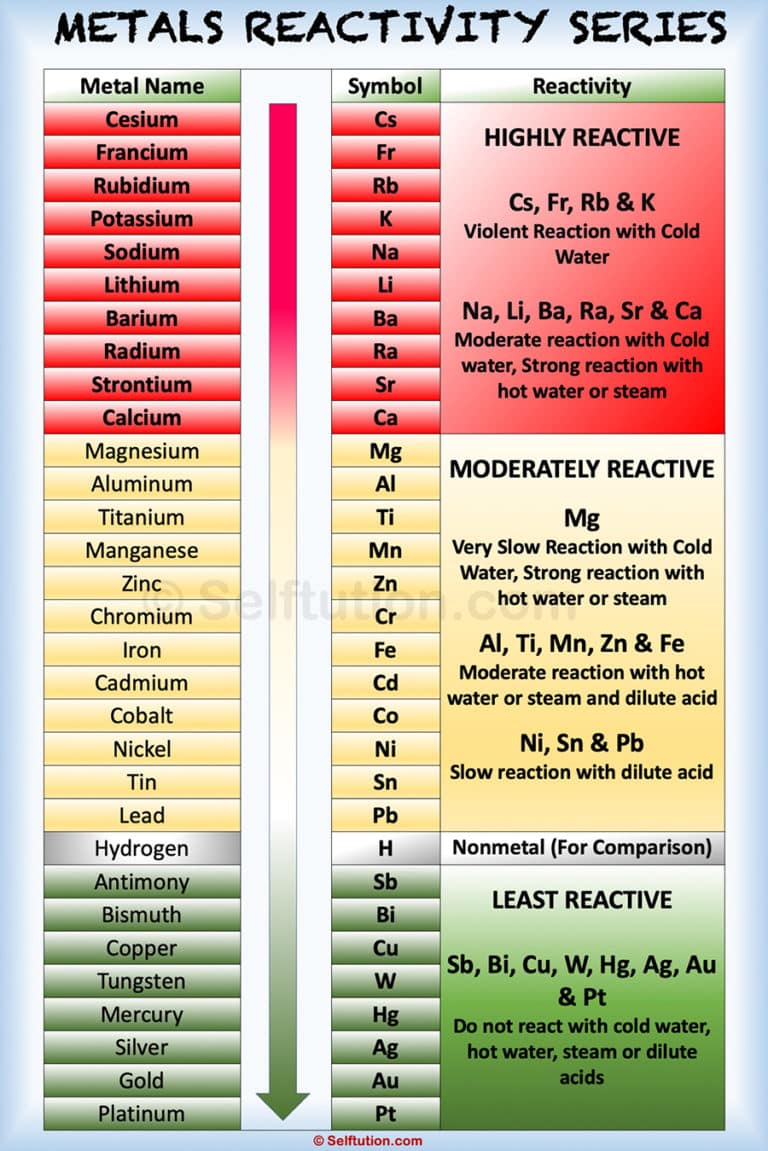
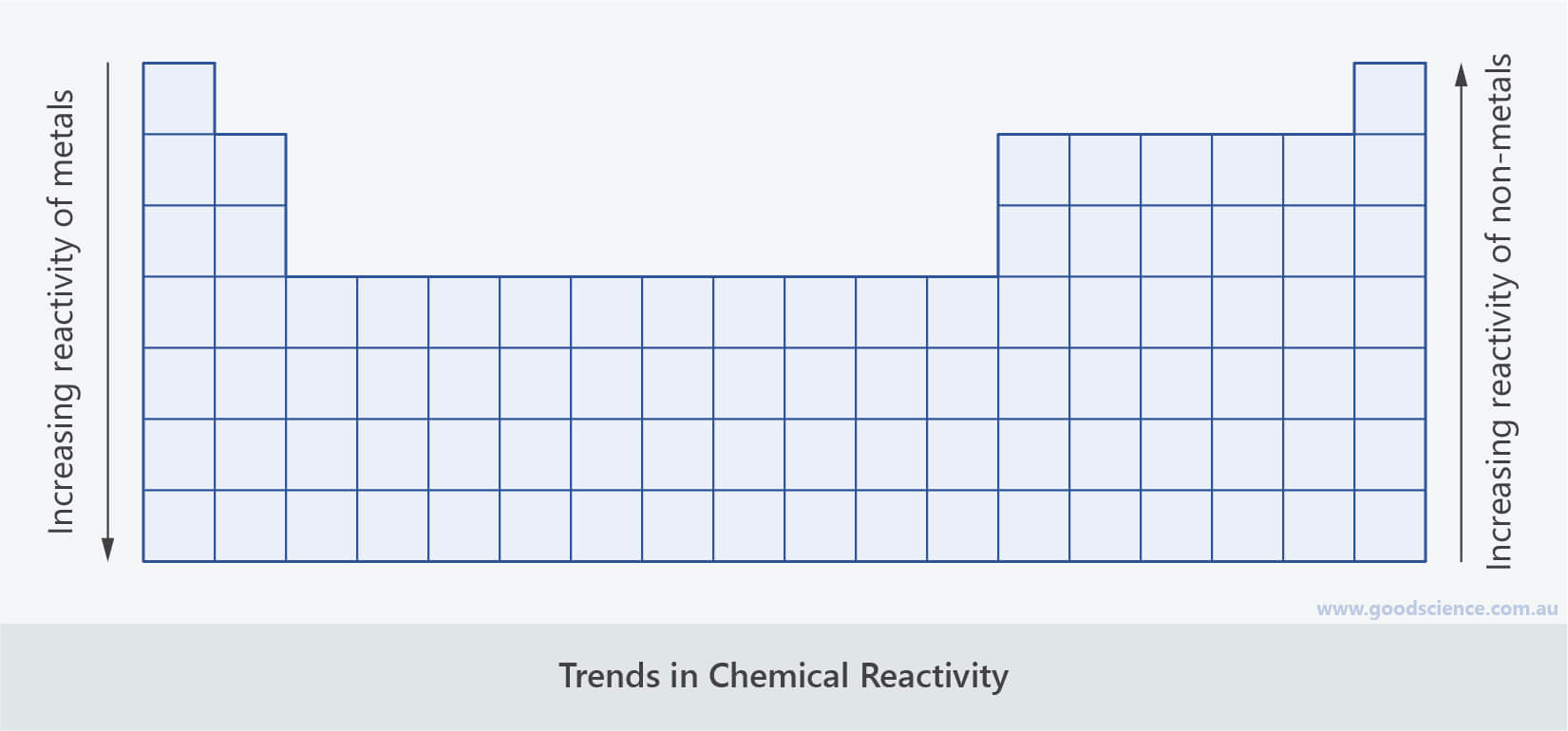
This generally correlates with electronegativity. the s- and p- sub-shells in the valence shell. The decrease in size from left to right across the table’s columns is due to how open the shells are, esp.The increase in size from row to row is due to the increased number of protons and neutrons in the nucleus as well as the corresponding increase in the number of electrons.Therefore, the smallest atom is Helium (Row 1, Column 18 of the standard Periodic Table), while the largest atom is Francium (Row 7, Column 1). The smallest sized atoms are in those with a complete valence shell (typically, this is s2p6, however in Period 1, there is only s2, which is why Helium behaves like a “noble gas”). Atomic sizes are largest in column one of the Periodic Table.NB: there are, in fact, more isotopes than this ranging from Iron-54 to Iron-60. It is easy to determine that there must be at least isotopes of Iron weighing 55 or 56 by subtracting the number of protons from the integer weights below or above the fractional number. Even though the actual distribution of isotopes may be unknown to you, the atomic weight being fractional will indicates that there must be more than one isotope of iron. Iron’s atomic weight is 55.85, while its atomic number is 26. The atomic weight reflects the isotopes of an element and their relative occurrence.The atomic number of an element is the number of protons in the nucleus Hydrogen 1, Helium 2, Lithium 3, etc.See original at Ĭhemistry becomes simpler to understand with the Periodic Table. Electron Energy Levels credit: Bruce Blaus This file is licensed under the Creative Commons Attribution 3.0 Unported license no changes were made. Gallium was discovered in 1875 and matched Mendeleev’s predictions – substitute Ga for Ea in the compounds mentioned above. In 1871 he predicted Eka-Aluminum would have an atomic mass of 68 (actual 69.7), a density of 6 gm/cm³ (actual 5.9), a low melting point (actual 29.8☌), and compounds with Oxygen or Chlorine. Mendeleev labeled an unknown element “Eka-Aluminum” because it would fill a gap in the same column as Aluminum but one row down. He could even make predictions about the properties of the undiscovered elements. Thus, he was able to leave gaps for elements, which were unknown at the time. Mendeleev recognized properties that repeated in a predictable, periodic pattern related to an element’s number of electrons. He then proceeded through the known elements sorting by weight and electrons. Hydrogen was the lightest element known, and it had one electron, so it became Element 1 on the Periodic Table. If Mendeleev did not know about protons and neutrons in the atomic nucleus, why does his Periodic Table work? Mendeleev understood the relationship between atomic weights and valence electrons, which gave him an indirect understanding of protons in the nucleus. Note that there is no clear division between metallic and non-metallic character.Learn more about elements: The Secret Lives of the Elements Why does Mendeleev’s Periodic Table Work? Fluorine gas reacts explosively with many other elements and compounds, and is considered to be one of the most dangerous known substances. It is not found in nature as a free element. Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The most reactive nonmetals reside in the upper right portion of the periodic table. 
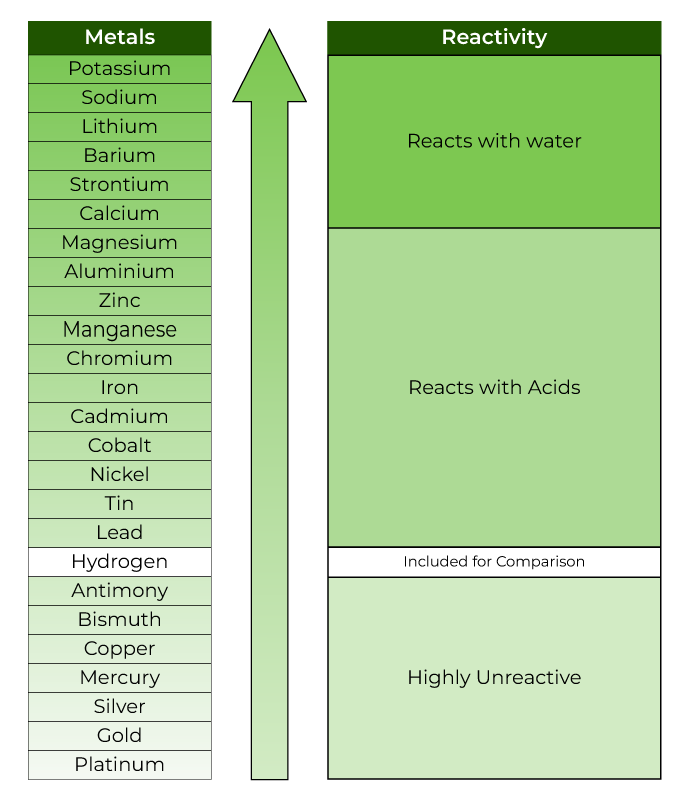
Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. In addition, the atomic radius increases going down a group, placing the outer electrons further away from the nucleus and making that electron less attracted by the nucleus. Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. The metallic character increases as you go down a group. Reactivity of metals is based on processes such as the formation of halide compounds with halogens, and how easily the element displaces hydrogen from dilute acids. (Credit: User:Mirek2/Wikimedia Commons Source: Commons Wikimedia, Periodic Trends(opens in new window) License: Public Domain)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
